 |
市場調查報告書
商品編碼
1273411
NGS 樣品製備市場增長、趨勢和預測 (2023-2028)NGS Sample Preparation Market - Growth, Trends, and Forecasts (2023 - 2028) |
||||||
※ 本網頁內容可能與最新版本有所差異。詳細情況請與我們聯繫。
在預測期內,NGS 樣本製備市場預計將以 12.4% 的複合年增長率增長。
COVID-19 大流行的爆發正在影響 NGS 樣本製備市場。 世界範圍內冠狀病毒感染人數不斷增加,因此有必要開發有效且快速的測序技術,以重建 SARS-CoV-2 的基因組序列,SARS-CoV-2 是 COVID-19 的病原體。我是。 它不僅在開發診斷性分子測試方面很重要,而且在製定有效的策略和戰略以減緩大流行的傳播方面也很重要。 例如,2020 年 8 月,Helix Laboratories 獲得了美國 FDA 緊急使用授權,可以進行基於 NGS 的 COVID-19 測試,以檢測上呼吸道標本中的 SARS-CoV-2 刺突蛋白基因。 這種下一代測序儀已應用於 COVID-19 研究,極大地促進了 SARS-CoV-2 的原始追蹤。
由於下一代測序 (NGS) 樣品製備在大流行中發揮了重要作用,市場經歷了正增長,預計在預測期內將繼續保持上升趨勢。
由於傳染病的流行和 NGS 平台的技術進步,測序成本下降是推動市場增長的主要因素。 隨著傳染病變得越來越普遍,NGS 測序技術已經進入病毒學領域,應用範圍很廣,包括從宏基因組樣本中檢測新病毒、重建整個或接近完整的病毒基因組序列,以及病毒進化和亞種分析. 選擇的方法正在迅速變化。 例如,根據 WHO 2022 年 8 月的流感更新,120 個國家、地區或區域國家流感中心 (NIC) 和其他國家流感實驗室已向 FluNet 提交了數據,直到 24 日。 在此期間,世衛組織全球流感監測和反應系統 (GISRS) 實驗室分析了超過 145,086 個樣本。 在流感病毒檢測呈陽性的 6,449 人中,97.7% 患有甲型流感,2.3% 患有乙型流感。 相似地, 根據世界衛生組織 2021 年報告,全球每年估計有 2100 萬人感染傷寒。
此外,據疾病預防控制中心稱,黃熱病每年影響 30,000 人,其中 90.0% 發生在非洲。 因此,預計在預測期內,全球傳染病數量的增加將推動市場增長。
主要和小型參與者發布的新 NGS 技術推動了市場增長。 NGS技術以其成本低、測序效率高等優點,已經取代了傳統的測序方法。 例如,據 Illumina 稱,自 2021 年人類基因組計劃完成以來,下一代測序 (NGS) 的成本已大幅下降。 Illumina 幫助降低了 NGS 的成本,使 1000 美元的人類基因組成為可能。 因此,預計新產品的推出將在預測期內推動市場增長。
然而,NGS 樣品製備設備的高成本以及與 NGS 樣品製備相關的嚴格法規可能會阻礙市場增長。
NGS 樣本製備的市場趨勢
NGS 可以快速對數百或數千個基因或整個基因組進行測序。
NGS 檢測的基因突變廣泛應用於疾病診斷、預後、治療決策和患者隨訪。
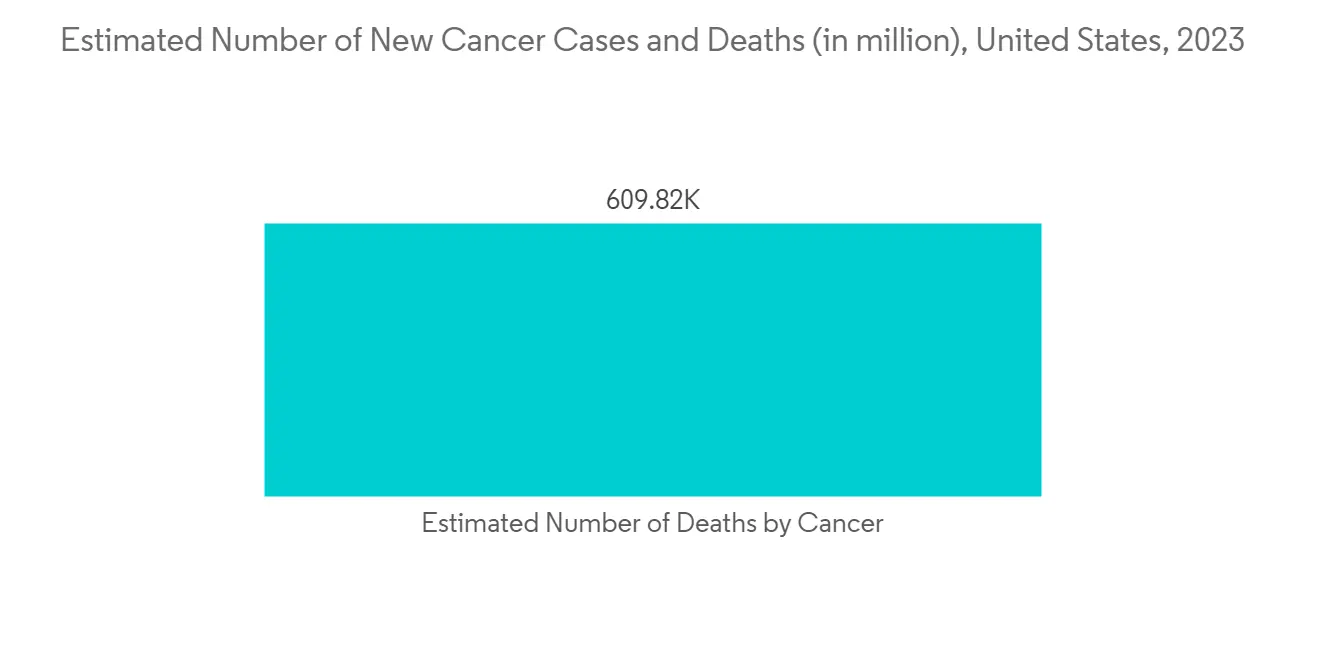
下一代測序極大地改變了癌症的研究和治療。 使用基於 NGS 的基因組測序對癌症患者的個體基因組進行 NGS,已成為最快、成本最低的方法之一。 例如,2021 年 8 月,腫瘤內科專業機構歐洲腫瘤學會 (ESMO) 發布了第一份關於對轉移性癌症患者使用下一代測序 (NGS) 的建議。 根據 ESMO 的建議,NGS 可能是結直腸癌中聚合□鏈反應 (PCR) 的替代方法。 通過對數百萬個 DNA 讀數進行測序,NGS 非常適合同時識別多個基因及其突變,以促進治療決策。 因此,用於藥物開發的下一代測序樣品製備的採用有望增加。
技術進步、主要參與者增加的產品批准和發布、合作夥伴關係和協作也在推動診斷行業的增長。 例如,2021 年 2 月,QIAGEN 和 INOVIO 擴大了合作,使用 INOVIO 的 VGX-3100 下一代測序 (NGS) 為晚期宮頸發育不良創建伴隨診斷。 此外,2020 年 8 月,Guardant Health Inc .獲得了美國食品藥品監督管理局(FDA)對 Guardant360 CDx 的批准,該產品在一項診斷測試中使用了液體活檢和 NGS 兩種組合技術。 因此,增加與 NGS 診斷相關的產品批准和產品發布可能會為該行業帶來新的機遇,從而在預測期內實現大幅增長。
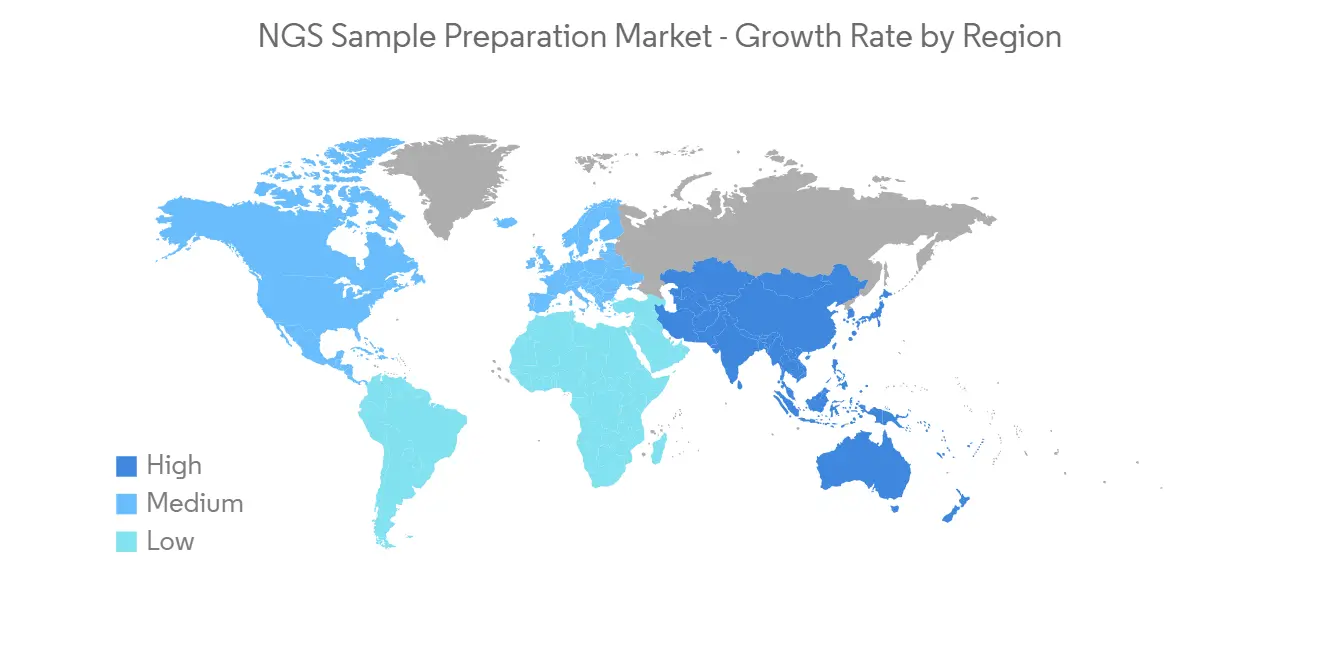
預計在預測期內北美將顯著增長
北美市場預計將經歷顯著的市場增長,原因是該地區對用於識別健康差異的診斷工具的需求不斷增長,以及傳染病和慢性病的負擔不斷增加。
主要市場參與者的技術進步、產品發布、合作夥伴關係和收購的增加導致北美市場增長加快。 例如,2022 年 1 月,Illumina 與范德比爾特大學醫學中心 (VUMC) 的子公司 Nashville Biosciences LLC 宣布,利用 Illumina 的新一代測序 (NGS) 平台進行大規模基因組分析,並建立臨床基因組資源,將引領簽訂多年協議以加速藥物開發。 這些努力有望推動北美 NGS 樣品製備市場的增長。
主要產品發布、集中的市場參與者和製造商存在、主要參與者之間的收購和合作夥伴關係、來自聯邦和私人參與者的資金增加、美國非政府和政府機構對 NGS 技術的採用是推動 NGS 增長的因素之一該國的 NGS 樣品製備市場。 例如,2021 年 4 月,安捷倫科技收購了 Resolution Biosciences,後者是基於下一代測序 (NGS) 的精準腫瘤學解決方案研究和商業化的領導者。 同樣,2020 年 1 月,美國情報高級研究計劃活動向布羅德研究所、哈佛大學和 DNA Scripts 撥款 2300 萬美元。 這兩個組織已經合作了四年多,以探索將□促 DNA 合成技術和 NGS 整合到一台儀器中的可能性。 因此,這種積極的發展預計將在預測期內推動美國市場的增長。
NGS樣品製備行業概覽
由於在全球和區域運營的參與者數量較少,NGS 樣本製備市場略有整合。 競爭格局包括對具有市場份額的知名國際和本地參與者的分析,包括 Illumina, Inc.、Agilent Technologies, Inc.、Bio-Rad Laboratories, Inc. 和 Thermo Fisher Scientific Inc.、Qiagen N.V.、PerkinElmer Inc.、 F.Hoffmann-La Roche Ltd、BGI Genomics、DNASTAR、Eurofins Scientific、Danaher Corporation (Beckman Coulter)、Macrogen Inc.、Integrated DNA Technologies, Inc. (Swift Biosciences Inc.)、Genomatix 和 Helix OpCo, LLC。
其他福利:
- Excel 格式的市場預測 (ME) 表
- 3 個月的分析師支持
內容
第一章介紹
- 調查假設和市場定義
- 本次調查的範圍
第二章研究方法論
第 3 章執行摘要
第四章市場動態
- 市場概覽
- 市場驅動因素
- 傳染病發病率上升
- 降低測序成本
- NGS 平台的技術進步
- 市場製約因素
- NGS 樣品製備設備成本高
- 與 NGS 樣品製備相關的嚴格規定
- 波特的五力分析
- 新進入者的威脅
- 買方/消費者議價能力
- 供應商的議價能力
- 替代品的威脅
- 競爭公司之間的敵對關係
第 5 章市場細分(市場規模、價值)
- 按產品類別
- 試劑和耗材
- 工作站
- 通過申請
- 診斷
- 藥物發現
- 其他用途
- 最終用戶
- 醫院和診所
- 製藥和生物技術公司
- 其他最終用戶
- 地區
- 北美
- 美國
- 加拿大
- 墨西哥
- 歐洲
- 德國
- 英國
- 法國
- 意大利
- 西班牙
- 其他歐洲
- 亞太地區
- 中國
- 日本
- 印度
- 澳大利亞
- 韓國
- 其他亞太地區
- 中東和非洲
- 海灣合作委員會
- 南非
- 其他中東和非洲地區
- 南美洲
- 巴西
- 阿根廷
- 其他南美洲
- 北美
第六章競爭格局
- 公司簡介
- Illumina, Inc.
- Agilent Technologies, Inc.
- Bio-Rad Laboratories, Inc.
- Thermo Fisher Scientific Inc.
- Qiagen N.V.
- PerkinElmer Inc.
- F. Hoffmann-La Roche Ltd
- BGI Genomics
- DNASTAR
- Eurofins Scientific
- Danaher Corporation(Beckman Coulter)
- Macrogen Inc.
- Integrated DNA Technologies, Inc.(Swift Biosciences Inc.)
- Genomatix
- Helix OpCo, LLC.
第七章市場機會與未來趨勢
The NGS sample preparation market is expected to register a CAGR of 12.4% over the forecast period.
The outbreak of the COVID-19 pandemic has impacted the NGS sample preparation market. The increasing coronavirus cases worldwide led to the need to develop effective and quick sequencing technologies to rebuild the genomic sequence of SARS-CoV-2, which was the etiological agent of COVID-19. It was critical in the development of diagnostic molecular tests as well as the development of efficient tactics and strategies to slow the spread of the pandemic. For instance, in August 2020, Helix Laboratory received the U.S. FDA emergency use authorization for NGS based COVID-19 test, which is designed to detect the SARS-CoV-2 spike protein gene in upper respiratory specimens. This next-generation sequencing was applied
for the study of COVID-19 and has greatly promoted SARS-CoV-2 original tracking. Since next-generation sequencing (NGS) sample preparation played a vital role during the pandemic, thus the market witnessed positive growth and is expected to continue the upward trend over the forecast period.
The rising infectious disease prevalence reduced sequencing cost, and technical advancement in NGS platforms are the major factors propelling the market growth. With the increasing prevalence of infectious diseases, NGS sequencing technologies have swiftly become the method of choice in virology for a wide range of applications, including the detection of novel viruses from metagenomic samples, reconstruction of whole or almost complete viral genome sequences, and viral evolution and subspecies analysis. For instance, as per the WHO August 2022 Influenza Update, National Influenza Centers (NICs) and other national influenza laboratories from 120 countries, regions, or territories submitted data to FluNet from 11 July 2022 to 24 July 2022. Over 145,086 samples were analyzed by the WHO Global Influenza Surveillance and Response System (GISRS) laboratories during that time. Among the 6,449 people who tested positive for influenza viruses, 97.7% had influenza A, and 2.3% had influenza B.
Similarly, according to the WHO report 2021, each year, typhoid fever is estimated to affect 21 million people worldwide. Similarly, according to the CDC, 30,000 cases of yellow fever occur annually, out of which 90.0% occur in Africa. Therefore, increasing cases of infectious diseases across the globe is anticipated to propel market growth over the forecast period.
The key and small players' launch of new NGS technologies drive market growth. NGS technologies have replaced traditional sequencing methods because of the low cost and high sequencing efficiency. For instance, according to Illumina, in 2021, the cost of next-generation sequencing (NGS) has decreased dramatically since the completion of the Human Genome Project. Illumina has helped reduce the cost of NGS, enabling the USD 1000 human genome. Therefore, new product launches are expected to surge the market growth over the forecast period.
However, the high costs of NGS sample preparation equipment and strict regulation associated with NGS Sample preparation are likely to impede the market growth.
NGS Sample Preparation Market Trends
Diagnostics Segment is Expected to Witness Considerable Growth Over the Forecast Period
NGS can sequence hundreds and thousands of genes or whole genomes quickly. The sequence variants/mutations detected by NGS have been widely used for disease diagnosis, prognosis, therapeutic decision, and follow-up of patients.
The research and treatment of cancer have been transformed by next-generation sequencing to a great extent. The NGS of individual cancer-patient genomes is carried out using NGS base genomic sequencing, and it has emerged as one of the fastest and less expensive methods. For instance, in August 2021, the European Society for Medical Oncology (ESMO), the medical oncology professional organization, launched its first recommendations on the use of next-generation sequencing (NGS) for patients with metastatic cancers. According to ESMO recommendations, NGS could be an alternative to Polymerase Chain Reaction (PCR) in colon cancers. NGS can be suitable for identifying multiple genes and their mutations concurrently by sequencing millions of DNA reads and driving therapeutic decision-making. This is projected to increase in adoption of next-generation sequencing sample preparation for drug development.
The advancements in technology, increasing product approvals and launches, partnerships, and collaborations by key players are also driving growth in the diagnostics segment. For instance, in February 2021, QIAGEN and INOVIO expanded their partnership to create a companion diagnostic using next-generation sequencing (NGS) for INOVIO's VGX-3100 for advanced cervical dysplasia. Furthermore, in Aug 2020, Guardant Health Inc. received the United States Food and Drug Administration (FDA) approval for Guardant360 CDx, which uses two combined technology, Liquid biopsy, and NGS, in one diagnostic test. Hence, the increasing product approval related to NGS diagnostic and product launches may create new opportunities for the segment, owing to which considerable segment growth is anticipated over the forecast period.

North America is Expected to Witness Significant Growth Over the Forecast Period
North America is expected to witness considerable market growth owing to factors such as the rising need for diagnostics tools for identifying health disparities, and the rising burden of infectious diseases and chronic diseases in the region. The increasing technological advancement, product launches, partnerships, and acquisitions by the key market players are leading to an increase in market growth in North America. For instance, in January 2022, Illumina Inc. and Nashville Biosciences LLC, a subsidiary of Vanderbilt University Medical Center (VUMC), entered a multi-year agreement to accelerate medicines development through large-scale genomics and the establishment of a clinical-genomic resource using Illumina's next-generation sequencing (NGS) platforms. Such initiatives are expected to drive the growth of the NGS sample preparation market in North America.
Key product launches, high concentration of market players or manufacturer's presence, and acquisition & partnerships among major players, and increased funding from the federal government and private players, as well as increased adoption of NGS technology by non-government and government bodies in the United States, are some of the factors driving the growth of the NGS sample preparation market in the country. For instance, in April 2021, Resolution Bioscience, a leader in the research and commercialization of next-generation sequencing (NGS)-based precision oncology solutions, has been acquired by Agilent Technologies. Similarly, in January 2020, in the United States, the Intelligence Advanced Research Projects Activity provided USD 23.0 million to the Broad Institute and Harvard University, and DNA Script. The organizations have been working together to explore the possibility of combining the enzymatic DNA synthesis technology and NGS into a single instrument for more than four years. Therefore, such positive developments are anticipated to boost the market growth in the United States over the forecast period.

NGS Sample Preparation Industry Overview
The NGS sample preparation market is slightly consolidated due to the presence of a few players operating globally and regionally. The competitive landscape includes an analysis of a few international as well as local companies which hold market shares and are well known, including Illumina, Inc., Agilent Technologies, Inc., Bio-Rad Laboratories, Inc., Thermo Fisher Scientific Inc., Qiagen N.V., PerkinElmer Inc., F.Hoffmann-La Roche Ltd, BGI Genomics, DNASTAR, Eurofins Scientific, Danaher Corporation (Beckman Coulter), Macrogen Inc., Integrated DNA Technologies, Inc. (Swift Biosciences Inc.), Genomatix, and Helix OpCo, LLC among others.
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 Study Assumptions and Market Definition
- 1.2 Scope of the Study
2 RESEARCH METHODOLOGY
3 EXECUTIVE SUMMARY
4 MARKET DYNAMICS
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Rising Prevalence of Infectious Diseases
- 4.2.2 Reduced Cost of Sequencing
- 4.2.3 Technical Advancement in NGS Platforms
- 4.3 Market Restraints
- 4.3.1 High Costs of NGS Sample Preparation Equipment
- 4.3.2 Strict Regulation Associated with NGS Sample Preparation
- 4.4 Porter's Five Force Analysis
- 4.4.1 Threat of New Entrants
- 4.4.2 Bargaining Power of Buyers/Consumers
- 4.4.3 Bargaining Power of Suppliers
- 4.4.4 Threat of Substitute Products
- 4.4.5 Intensity of Competitive Rivalry
5 MARKET SEGMENTATION (Market Size by Value - USD Million)
- 5.1 By Product Class
- 5.1.1 Reagent and Consumables
- 5.1.2 Workstations
- 5.2 By Applications
- 5.2.1 Diagnostics
- 5.2.2 Drug Discovery
- 5.2.3 Other Applications
- 5.3 End User
- 5.3.1 Hospitals and Clinics
- 5.3.2 Pharmaceutical and Biotechnology Companies
- 5.3.3 Other End Users
- 5.4 Geography
- 5.4.1 North America
- 5.4.1.1 United States
- 5.4.1.2 Canada
- 5.4.1.3 Mexico
- 5.4.2 Europe
- 5.4.2.1 Germany
- 5.4.2.2 United Kingdom
- 5.4.2.3 France
- 5.4.2.4 Italy
- 5.4.2.5 Spain
- 5.4.2.6 Rest of Europe
- 5.4.3 Asia-Pacific
- 5.4.3.1 China
- 5.4.3.2 Japan
- 5.4.3.3 India
- 5.4.3.4 Australia
- 5.4.3.5 South Korea
- 5.4.3.6 Rest of Asia-Pacific
- 5.4.4 Middle East and Africa
- 5.4.4.1 GCC
- 5.4.4.2 South Africa
- 5.4.4.3 Rest of Middle East and Africa
- 5.4.5 South America
- 5.4.5.1 Brazil
- 5.4.5.2 Argentina
- 5.4.5.3 Rest of South America
- 5.4.1 North America
6 COMPETITIVE LANDSCAPE
- 6.1 Company Profiles
- 6.1.1 Illumina, Inc.
- 6.1.2 Agilent Technologies, Inc.
- 6.1.3 Bio-Rad Laboratories, Inc.
- 6.1.4 Thermo Fisher Scientific Inc.
- 6.1.5 Qiagen N.V.
- 6.1.6 PerkinElmer Inc.
- 6.1.7 F. Hoffmann-La Roche Ltd
- 6.1.8 BGI Genomics
- 6.1.9 DNASTAR
- 6.1.10 Eurofins Scientific
- 6.1.11 Danaher Corporation (Beckman Coulter)
- 6.1.12 Macrogen Inc.
- 6.1.13 Integrated DNA Technologies, Inc. (Swift Biosciences Inc.)
- 6.1.14 Genomatix
- 6.1.15 Helix OpCo, LLC.











